The United Kingdom has confirmed that it will deploy "resuscitation facilities" in coronavirus vaccination centers. The National Health Service (NHS) confirmed the move Dec. 9 after two healthcare workers suffered allergic reactions, following their immunization with the Pfizer/BioNTech vaccine on the first day of its distribution the day before. Pfizer's BNT162b2 vaccine made in partnership with German company BioNTech was the first jab against the Wuhan coronavirus approved in the country.
In line with the adverse reactions, the Medicines and Healthcare products Regulatory Agency (MHRA) issued "precautionary advice" discouraging people who have a "history of significant reaction" to medicines, foods or vaccines from taking the COVID-19 vaccine. An earlier NHS safety warning advised pregnant and lactating women to avoid the vaccine as its effects on fertility are "unknown."
The new NHS advice stated: "Resuscitation facilities should be available at all times for all vaccinations. Vaccination should only be carried out in facilities where resuscitation measures are available. NHS England and NHS Improvement spokesman Matthew Edwards told Breitbart London Dec. 8 that "vaccination hubs all follow the guidelines set by the MHRA."
Meanwhile, the MHRA did not respond to Breitbart London's request for comment by the time of publication.
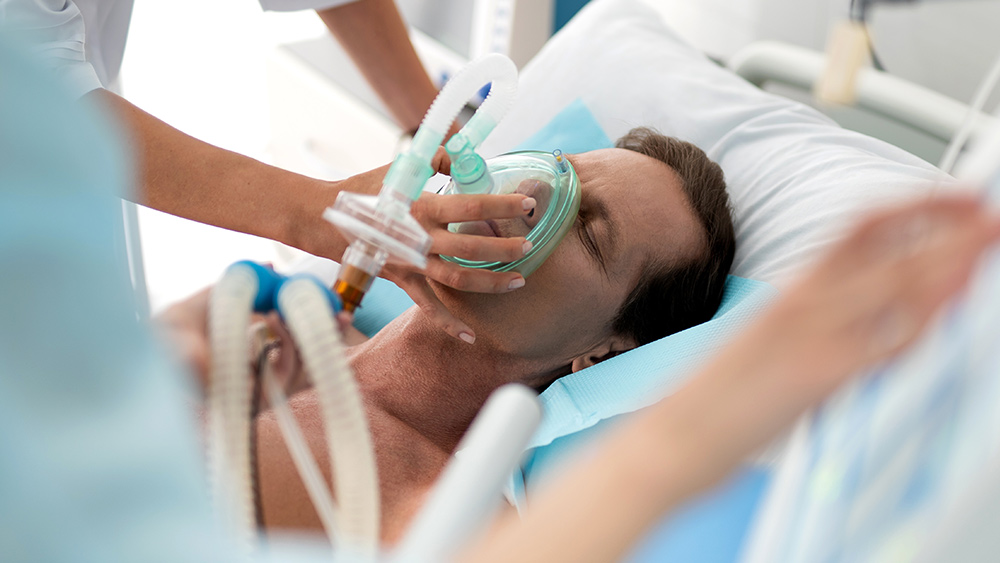
The new rules followed two healthcare workers who received the vaccine experiencing anaphylactic reactions; both recovered after receiving medical treatment. Incidentally, the health workers carried adrenaline autoinjectors to deal with their allergies.
Based on Johns Hopkins University data, the U.K. currently has a 1.7 million COVID-19 caseload with 3,786 recoveries and 63,179 deaths.
Professors have said that allergic reactions are common with vaccines such as BNT162b2
The NHS describes anaphylaxis as a "severe and potentially life-threatening reaction to a trigger such as an allergy" caused by certain foods, medicines and chemicals.
Pharmacoepidemiology professor Stephen Evans of the London School of Hygiene and Tropical Medicine said allergic reactions are not unexpected as they occur "with quite a number of vaccines, and perhaps even more frequently with drugs." The professor continued that hypersensitivity to the vaccine itself or its ingredients is a valid reason to avoid the jab, but some people would not know if they are hypersensitive to some of the vaccine's ingredients.
Thus, Evans advised those who have a history of allergies and those who need to carry an autoinjector to put off taking the vaccine "until the reason for the allergic reaction has been clarified."
Imperial College London experimental medicine professor Peter Openshaw said: "As with all food and medications, there is a very small chance of an allergic reaction to any vaccine. However, it is important that we put this risk in perspective." (Related: Pfizer boss warns against fast-tracking vaccines.)
Openshaw continued that the MHRA is monitoring the new COVID-19 vaccine and will perform an investigation to understand if the allergic reactions experienced by the two healthcare workers were linked to the vaccine or were incidental. He commented: "The fact that we know so soon about these two allergic reactions and that the regulator has acted on this to issue precautionary advice shows that this monitoring system is working well."
It's not just Pfizer: Other coronavirus vaccine candidates have caused adverse reactions
Prior to the U.K. approving the Pfizer/BioNTech vaccine for emergency use, some volunteers who participated in COVID-19 vaccine trials by other drugmakers experienced adverse reactions.
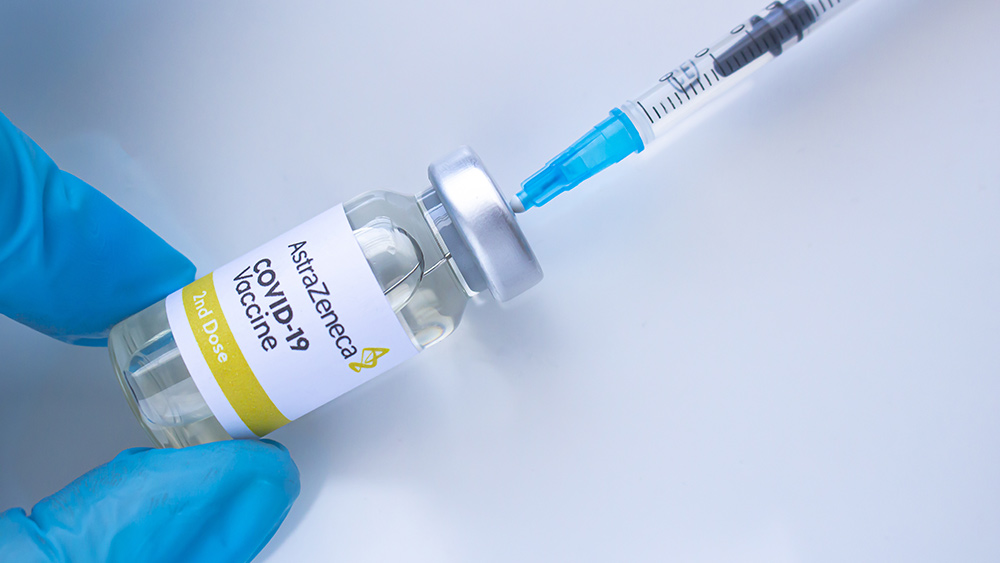
British firm AstraZeneca reported two volunteers experienced spinal inflammation after being injected with its experimental COVID-19 vaccine. The first reaction was documented in July, while another adverse instance was recorded in September. A month later, one Brazilian participant died during phase three trials in the South American nation.
AstraZeneca's vaccine developed in partnership with the University of Oxford makes use of a harmless adenovirus. This adenovirus – similar to the one that causes the common cold – triggers the body to produce antigens that provide immunity to the Wuhan coronavirus. However, further scrutiny found that toxic ingredients in the vaccine candidate such as aborted human fetal tissue had a role in causing the serious reactions in the three victims.
Despite these red flags, the British firm still insisted on publishing that its vaccine candidate demonstrated a 90 percent efficacy rate – which was caused by a mistakenly administered one-and-a-half doses. (Related: AstraZeneca's COVID-19 vaccine will "never be licensed in the US," says analyst.)
Stay updated with the latest news about Wuhan coronavirus vaccines at Vaccines.news.
Sources include:
Please contact us for more information.