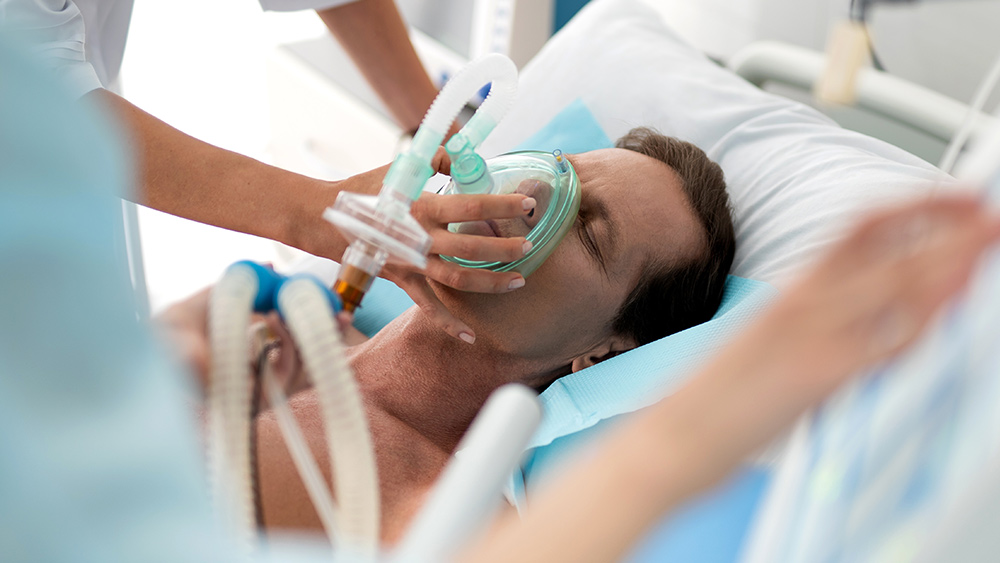
A group of 20 Republican senators sent a letter to the Food and Drug Administration this week asking them to “classify the abortion pill as an ‘imminent hazard to the public health’ that poses a ‘significant threat of danger’ and remove this pill from the U.S. market.”
Mifepristone is the first drug given to women in a medical abortion, which occurs when a woman ends a pregnancy by taking pills instead of undergoing surgery. The process usually entails an initial dose of 200 milligrams of mifepristone, which will stop progesterone from going to the fetus. This is followed 24 to 48 hours later by 800 grams of misoprostol, which is either inserted between the cheeks and gums or vaginally and prompts the uterus to contract and empty.
In the letter, the senators, who were led by Sen. Ted Cruz of Texas, said that the pill presents a greater risk of complications than surgical abortion in early pregnancy. The pill can cause vomiting, cramping, heavy bleeding and even loss of consciousness. As many as 7 percent of women end up needing follow-up surgical abortions after all. Thousands of women are harmed even by using the drug correctly, not to mention the millions of children who are killed by it.
They cited FDA statistics showing that more than 3.7 million preborn lives have been lost to the abortion pill. It has caused 24 maternal deaths and more than 4,195 adverse maternal reactions such as life-threatening infections, severe abdominal pain and hemorrhage. However, many of these adverse events go unreported, so the true number could be significantly higher.
The senators applauded the FDA for putting safety protocols in place related to the chemical abortion pill, which is also known as Mifeprex or RU-486. Their letter also noted how President Clinton rushed the pill through the approval process because of political pressure. They added that the accelerated approval process it was given is normally reserved for risky drugs that address life-threatening illnesses.
“As you are surely aware, pregnancy is not a life-threatening illness, and the abortion pill does not cure or prevent any disease,” they wrote in the letter.
Women no longer need to see a doctor in person to receive the abortion pill
Their letter also referred to a recent federal district court order that eliminates the in-person requirements of the FDA’s Risk Evaluation and Mitigation Strategies, or REMS. These are formal plans the FDA has put in place to determine if the benefits will outweigh the risks before people take high-risk drugs. Under REMS, mifepristone can only be given in doctor’s offices, clinics or hospitals in person.
The providers of the pill must be able to accurately date the pregnancy as it can only be used within 70 days of a woman’s last menstrual period. They also need to be able to recognize a potential ectopic pregnancy and arrange for a surgical abortion should it be necessary if the pill fails.
The abortion industry took advantage of the coronavirus pandemic to push for the potentially life-saving measures of REMS to be set aside given the difficulty some women might have in accessing in-person care. The senators said in their letter that this decision, which was handed down by U.S. District Judge Theodore Chuang, was a case of “rogue judicial activism” that undermined the authority of the FDA and “recklessly endangered” women and children. Women who are given the pill via telemedicine are unlikely to get the necessary screening, blood tests and ultrasounds to determine if it is safe to proceed.
The letter pointed out that by demedicalizing and deregulating the pill, many women end up carrying out “DIY” chemical abortions that put their lives in danger while the abortion industry racks up profits. Women deserve to know that these pills are putting their lives in danger.
Sources for this article include:
Please contact us for more information.